Fluorescence-based techniques have become fundamental tools in modern biological and biomedical research. From visualizing cellular structures to tracking molecular interactions, fluorescence imaging enables scientists to observe biological processes with remarkable precision. At the center of these techniques are fluorophores, also known as fluorescent dyes, which emit light when excited by specific wavelengths.
However, selecting the right fluorophore for an experiment is not always straightforward. Researchers must consider multiple factors including excitation and emission spectra, spectral overlap, brightness, photostability, and compatibility with imaging platforms such as confocal microscopy. These factors become even more critical in multiplex fluorescence imaging, where multiple fluorescent probes are used simultaneously.
This article provides a practical fluorophore selection guide to help researchers choose the most suitable dyes for their experiments, while highlighting widely used fluorophores such as Alexa Fluor dyes, FITC, Cy3, Cy5, DAPI, ATTO dyes, and quantum dots.
What Are Fluorophores?
Fluorophores are molecules that absorb light energy at one wavelength and emit light at a longer wavelength. This process, known as fluorescence, occurs when a molecule absorbs photons, becomes excited to a higher energy state, and then releases part of that energy as emitted light.
In biological research, fluorophores are commonly attached to molecules such as antibodies or nucleic acids. When these labeled molecules bind to their targets, the fluorophore allows researchers to detect and visualize them using fluorescence microscopy or other imaging techniques.
Fluorophores are widely used in:
-
Immunofluorescence microscopy
-
Flow cytometry
-
Live-cell imaging
-
Protein localization studies
-
DNA and RNA detection
-
Multiplex fluorescence imaging
The success of these applications depends heavily on selecting the right fluorescent dye for the specific experimental setup.
Understanding Excitation and Emission Spectra
One of the most important considerations when choosing fluorophores is understanding their excitation and emission spectra.
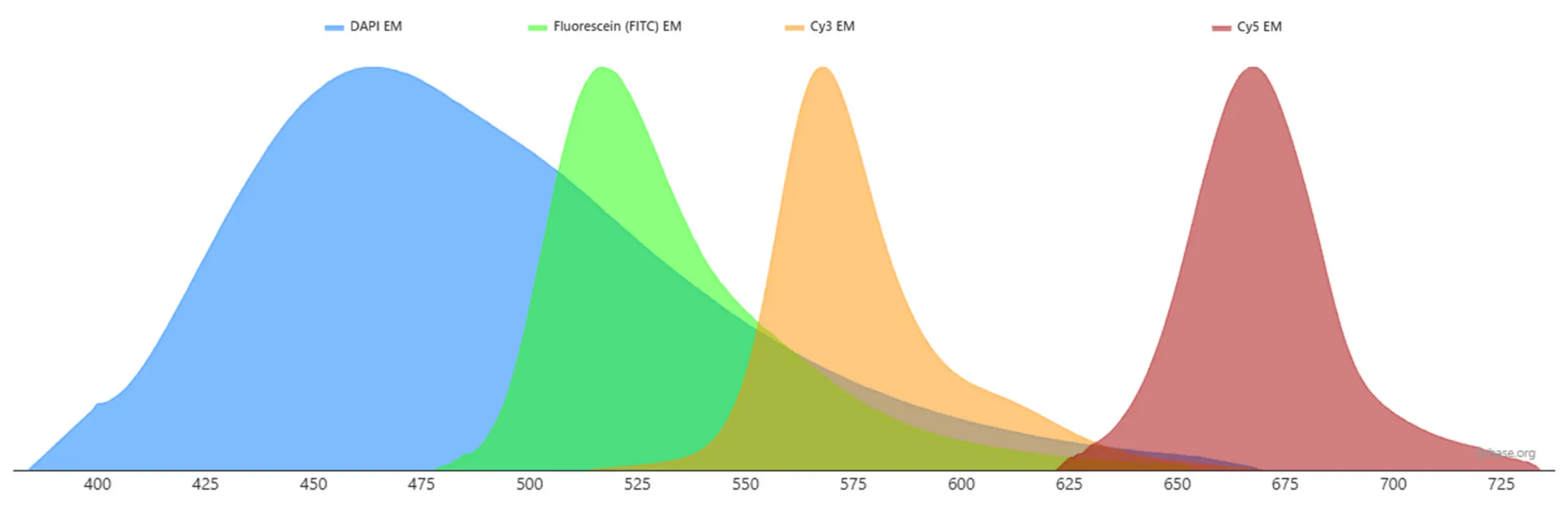
Each fluorophore has characteristic peak wavelengths for excitation and emission. For optimal performance, the excitation source—such as a microscope laser—should closely match the fluorophore’s excitation peak. Likewise, the emission detection filters should match the emission peak.
For example:
- FITC (Fluorescein isothiocyanate)
- Excitation peak: ~495 nm
- Emission peak: ~519 nm
- DAPI
- Excitation peak: ~358 nm
- Emission peak: ~461 nm
Understanding these spectral properties ensures that the fluorophore can be efficiently excited and accurately detected by the imaging system.
Avoiding Spectral Overlap in Multiplex Imaging
When experiments require labeling multiple targets simultaneously, researchers often use multiplex fluorescence imaging. In these experiments, several fluorophores are used in the same sample.
A key challenge in multiplex imaging is spectral overlap, which occurs when emission spectra from different fluorophores overlap significantly. This overlap can lead to signal bleed-through between channels, making it difficult to distinguish individual targets.
To minimize spectral overlap, researchers should choose fluorophores with well-separated emission wavelengths. A common multi-color panel might include:
-
DAPI – blue emission
-
FITC – green emission
-
Cy3 – orange emission
-
Cy5 – far-red emission
This spectral separation allows researchers to capture distinct signals from multiple biological targets without interference.
Advanced microscopes can also use spectral unmixing techniques, but proper fluorophore selection remains the most reliable way to avoid signal contamination.
Fluorophore Compatibility with Confocal Microscopy
In confocal microscopy, fluorophores are excited by specific laser lines. Therefore, fluorophore selection must be compatible with the available lasers in the microscope.
Common confocal laser wavelengths include:
-
405 nm
-
488 nm
-
561 nm
-
633–640 nm
Matching fluorophores to these lasers improves fluorescence signal intensity and imaging efficiency.
Examples of compatible pairings include:
Before selecting fluorophores, researchers should review the specifications of their microscope’s lasers and detection filters to ensure compatibility.
Common Fluorescent Dyes Used in Biological Research
Alexa Fluor Dyes
Alexa Fluor dyes are among the most widely used fluorophores in fluorescence microscopy and immunostaining experiments. These dyes are known for their exceptional brightness and high photostability.
Key advantages include:
Because of these properties, Alexa Fluor dyes are commonly used for antibody labeling and high-resolution imaging experiments. FITC
FITC is one of the earliest and most widely used fluorescent dyes in biological research. It emits bright green fluorescence and is commonly used in immunofluorescence and flow cytometry.
Although FITC is easy to use and cost-effective, it has relatively low photostability compared to newer dyes. Cyanine Dyes (Cy3 and Cy5)
Cyanine dyes such as Cy3 and Cy5 are widely used for multi-color fluorescence experiments.
-
Cy3 emits orange fluorescence.
-
Cy5 emits far-red fluorescence, making it easily separable from other common fluorophores and so ideal for multiplex imaging
These dyes are frequently used in applications such as DNA microarrays, fluorescence in situ hybridization (FISH), and antibody labeling. ATTO Dyes
ATTO dyes provide high brightness and excellent photostability, for biological imaging and analysis.ATTO dyes also exhibit good resistance to photobleaching, which is important for long imaging sessions or time-lapse experiments. DAPI
DAPI is a fluorescent stain that binds strongly to DNA, making it a standard dye for labeling cell nuclei. When bound to DNA, DAPI becomes much more strongly fluorescent than in free solution, which makes nuclei appear bright and easy to visualize. Because it emits bright blue fluorescence, DAPI is often used as a counterstain alongside other fluorophores in multicolor imaging experiments. Quantum Dots
Quantum dot probes represent a relatively newer class of fluorescent labels. These semiconductor nanoparticles exhibit extremely bright fluorescence and narrow emission spectra.
Advantages of quantum dots include:• Exceptional brightness• Very high photostability• Narrow emission spectra minimizes signal bleed-through
These properties make quantum dots useful for long-term imaging and multiplex experiments. However, quantum dots also have drawbacks, including their relatively large size, more complex surface chemistry, and potential toxicity concerns depending on composition.
Key Factors to Consider When Selecting Fluorophores
When designing a fluorescence experiment, researchers should evaluate several key parameters before selecting fluorophores.
Brightness
Brightness determines how easily a fluorophore can be detected. It depends on two main factors: the fluorophore’s extinction coefficient and quantum yield. Brighter fluorophores are especially important when detecting low-abundance targets.
Photostability
Photostability refers to a fluorophore’s resistance to photobleaching during illumination. Highly photostable dyes maintain their signal during long imaging sessions.
Spectral Separation
Choosing fluorophores with minimal spectral overlap is essential for multiplex experiments to ensure accurate signal detection.
Instrument Compatibility
Fluorophores must match the available excitation lasers, filter sets, and detectors in the microscope or imaging system. Example Fluorophore Panel for Multiplex Imaging
A typical four-color fluorescence experiment may include the following dyes:
| Target |
Fluorophore |
| Nucleus |
DAPI |
| Protein A |
Alexa Fluor 488 or FITC |
| Protein B |
Cy3 |
| Protein C |
Cy5 |
This combination provides good spectral separation and is compatible with most confocal microscopes.
Conclusion
Fluorophores are essential tools in modern biological imaging, enabling researchers to visualize cellular structures and molecular interactions with high precision. However, successful fluorescence experiments require careful fluorophore selection.
By considering factors such as excitation and emission spectra, spectral overlap, brightness, photostability, and fluorophore compatibility with lasers, researchers can design effective experiments and achieve high-quality imaging results.
Whether using traditional dyes like FITC and DAPI, advanced probes such as Alexa Fluor dyes and ATTO dyes, or technologies like quantum dots, choosing the right fluorescent dye is critical for reliable and reproducible fluorescence imaging.
Thoughtful fluorophore selection ultimately enables clearer images, more accurate data, and deeper insights into complex biological systems.
